With the emergence of antibiotic resistance, one goal of research scientists has been to understand through which medium bacteria communicate, and how we may inhibit this conversation.
Why? Biofilms and virulence. Biofilms are a growing concern, as they are largely responsible for antibiotic-resistance, acting as an impenetrable shield which protects pathogenic microcolonies from the affects of antimicrobials and the immune system, whilst supporting their adherence to a surface. Bacterial communication is essential for the assembly of bacteria microcolonies, subsequent biofilm formation and also activation of virulence factors (e.g. release of symptom-causing toxins, enzymes that degrade antimicrobial products, and allow disease spread).
If a therapy was able to block interactions between bacteria, it would hold the potential to suppress the biofilm-forming and virulence functions of these potentially devastating treatment-resistance pathogens.
But how? Bacterial communication, known as “quorum sensing” (QS) has been identified as the method through which bacteria are able to identify the presence of like-species (through specific quorum sensing molecules (QSM) that are unique to a bacterial species) and of other bacterial forms (through a different QSM found to be expressed by all bacteria).
Bacteria express certain QS receptors (QSR) which detect the presence of these QSM, while the bacteria persistently release QSM themselves. When bacteria numbers grow, the local concentration of QSM increases exponentially. This increased concentration is detected by QSR, which subsequently triggers the expression of specific transcription factors within the microorganism, ultimately “turning on” functions such as those involved in biofilm formation and virulence.
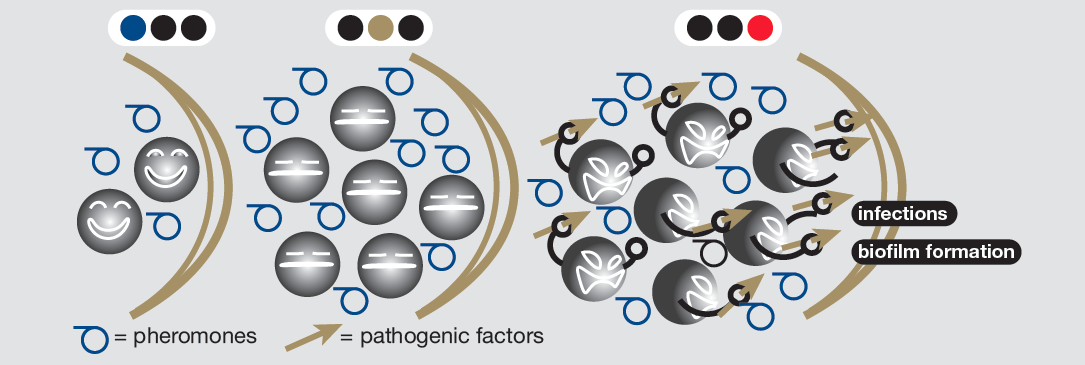
Cell dependent gene expression in quorum sensing (virulence expression)
Understanding that QSM are involved in ensuring these functions are triggered, it is possible that the ability to interfere with QSM release and QSR function could prevent and manage infections.
Phytochemicals, such as allicin from Allium sativum (garlic), have long been recognised for their antimicrobial benefits. Recent evidence suggests that this may, in part, be attributed to allicin’s inhibitory effects on QS. An in vitro study investigating the anti-biofilm and anti-virulence effects of allicin against Pseudomonas aeruginosa identified that the phytochemical was able to suppress certain QS-controlled virulence factors, including maintenance of biofilm structure, release of enzymes capable of degrading immunological agents, and toxins contributing to infection symptoms. It was suggested that these actions are exerted via modulation of QSM binding to QSR, and inhibition of certain aspects of QS gene expression (however more research is needed to elucidate exact mechanisms of action).[1]
Curcumin from Curcuma longa (turmeric) and 6-gingerol from Zingiber officinale (ginger) are also identified to exert QS suppression against P. aeroginosa. Curcumin appears to function by inhibiting virulence gene expression,[2] while 6-gingerol has demonstrated anti-biofilm and virulence-suppressing effects that are attributed to an antagonist function at QSR.[3]
Additional phytochemicals exerting actions which target QS include urolithin-A and B (these are the result of ellagic acid metabolism which is yielded from hydrolysis of ellagitanins (e.g. from Punica granatum (pomegranate) in the gut), rosmarinic acid (e.g. from Rosmarinus officinalis (rosemary), and Perilla frutescens (perilla)) and epigallocatechin gallate (EGCG) from Camellia sinensis (green tea).[2]
N-acetylcysteine (NAC) is also a potential suppressor of QS actions. It is suggested that this function is owed to the antioxidant capabilities of NAC. Reactive oxygen species seem to play a role in activating the quorum sensing pathways, which is inhibited by NAC’s ability to suppress hydrogen peroxide activity and the master quorum sensing regulators. This effect on hydrogen peroxide was also able to inhibit the toxin release from P. aeruginosa, as virulence factors such as toxin secretion are also stimulated by oxidative stress.[4]
Due to the varying actions exerted by different phytochemicals and nutrients (and due to the potentially unachievable doses required for a single ingredient), it is hypothesised that a broad-spectrum approach may be desirable when looking to optimise results. QS inhibitors may also be used in conjunction with other known biofilm disruptors (e.g. enzymes, probiotics), or pharmaceutical interventions.
Moreso, a preventative approach to chronic infection is ideal (particularly in those that are high-risk), with a diet rich in anti-QS phytochemicals recommended (e.g. in addition to those discussed, further useful actives can be found in commonly consumed plants including beansprouts, chamomile, lemongrass, carrots, yellow capsicum and seaweed).[5]
References
- Lihua L, Jianhuit W, Jialini Y, et al. Effects of allicin on the formation of Pseudomonas aeruginosa biofinm and the production of quorum-sensing controlled virulence factors. Pol J Microbiol. 2013;62(3):243-251. [Full text]
- Nazzaro F, Fratianni F, Coppola R. Quorum sensing and phytochemicals. Int J Mol Sci 2013;14(6):12607-12619. [Full text]
- Kim HS, Lee SH, Byun Y, et al. 6-Gingerol reduces Pseudomonas aeruginosa biofilm formation and virulence via quorum sensing inhibition. Sci Rep. 2015;5:8656. [Full text]
- Hasanin A, Omri A. Liposomal N-acetylcysteine modulates the pathogenesis of P. aeruginosa isolated from the lungs of cystic fibrosis patient. J Nanomed Nanotechnol 2014;5(4):1-12. [Full text]
- Givskov M. Beyond nutrition: health-promoting foods by quorum-sensing inhibition. Future Microbiol 2012;7(9):1025-1028. [Full text]
DISCLAIMER:
The information provided on FX Medicine is for educational and informational purposes only. The information provided on this site is not, nor is it intended to be, a substitute for professional advice or care. Please seek the advice of a qualified health care professional in the event something you have read here raises questions or concerns regarding your health.