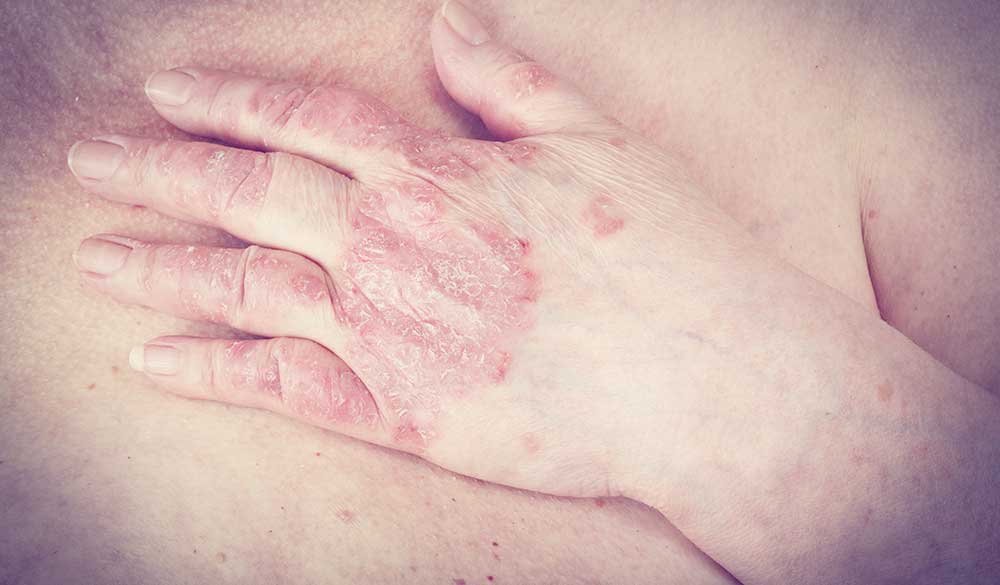
Psoriasis is a chronic inflammatory disease that largely affects the skin. It can also be associated with iritis and disturbances in other body systems, such as the gastrointestinal and musculoskeletal systems.
Globally, 125 million people are affected by psoriasis and approximately 30% of them are likely to go on to develop psoriatic arthritis (PsA).[1-3]
Like psoriasis, PsA is also chronic and inflammatory in nature. It is an autoimmune arthritis that forms part of the seronegative spondyloarthropathies, a group of diseases that also includes reactive arthritis, ankylosing spondylitis, enteropathic arthritis and an association with the HLA-B27 antigen. PsA tends to affect those between the ages of 40 to 50, however it can also develop in children and the elderly.[3-8]
Treatment for PsA is aimed at suppressing the autoimmune response, reducing inflammation, alleviating troublesome symptoms, reducing the ongoing damage to joint structures and improving patient quality of life.[5]
Background on PsA
PsA is an autoimmune condition that is characterised by the presence of both psoriasis and PsA. It is a chronic, typically asymmetrical, and a peripheral disease that predominantly presents with inflammation of small joints and surrounding structures. Other associated symptoms include morning stiffness, pain or tenderness over affected joints (sometimes with swelling), nail pitting and flaking, fatigue and difficulty performing general daily tasks. Symptoms may also include iritis and sluggish bowel function.
The pathogenesis of psoriasis and PsA is not thoroughly understood. However, several exogenous and endogenous factors are believed to contribute to both the onset and exacerbation of these conditions. These include environmental factors such as stress, emotional trauma, humidity and/or cold weather, certain drugs, streptococcal pharyngitis (with guttate psoriasis), obesity and smoking, along with immunologic and genetic factors.[2] Several studies also note that as many as 40% of patients with PsA have a concurrent psychiatric diagnosis.[8]
Five distinct subtypes of PsA are outlined:[6,7,9]
- Symmetric polyarthritis – May have symmetric involvement in five or more joints and can resemble rheumatoid arthritis.
- Asymmetric oligoarthritis – Involves five or fewer joints and is commonly seen in an asymmetric distribution.
- Distal interphalangeal predominant (joint involvement) – Tends to develop in conjunction with other subtypes and involves distal interphalangeal joints of the feet, hands, or both. Rarely occurs as a single condition.
- Arthritis mutilans – The most destructive of the subtypes that also results in deformity. This form of arthritis can cause significant bone resorption and destruction of bone tissue. It is often characterised by a compressing, overlapping or deformity of digits.
- Spondylotarthritis or axial arthritis – Largely involves both the spine and sacroiliac joints.
The prevalence of each PsA subtype can vary, but symmetric polyarthritis and asymmetric oligoarthritis are the most common.[3]
Increased risk of comorbid conditions
Studies have shown that patients with PsA are at a higher risk of cardiovascular disease (CVD), as the condition is correlated with atherosclerosis. There is also a higher prevalence of inflammation, dyslipidaemia, hyperglycaemia, obesity, metabolic syndrome and type 2 diabetes, ischaemic heart disease, angina and myocardial infarction, arterial hypertension, as well as heart failure and vascular disease.[10] Raised serum IgA antibodies to gliadin (AGA) and coeliac disease are also more prevalent in these patients.[11]
Furthermore, gut dysbiosis is likely to be present in PsA with populations of Akkermansia and Ruminococcus (important for homeostasis within the gut), Pseudobutyrivibrio and Coprococcus being significantly decreased. This was shown to correlate with increased inflammation and an increased concentration of secretory IgA along with a reduction of some medium chain fatty acids, thereby impacting both the intestinal ecosystem and permeability.[12]
Diagnosis
The diagnosis of PsA is made clinically using the classification of psoriatic arthritis (CASPAR) criteria. The criteria are considered met if inflammatory musculoskeletal disease with three or more of the following are present: negative rheumatoid factor, current psoriasis or a history of psoriasis, inflammation of an entire digit (finger or toe), psoriatic nail degeneration or radiographic evidence of new bone formation near joints.[13]
Current medical management
Medical treatment for PsA involves the use of several medications to manage the pain and debilitating symptoms associated with the condition. Corticosteroids, non-steroidal anti-inflammatory drugs (NSAIDs), conventional disease-modifying antirheumatic drugs (DMARDs) and antitumor necrosis factor agents are prescribed singularly or in combination to provide support to patients. Several considerable side-effects are routinely experienced with these medications,[3,5] which leaves patients looking for complementary or alternative options.
Naturopathic considerations and management
Natural therapies have had great effect in the management of PsA and treatment should aim to reduce inflammation, modulate the immune response, preserve the structure and movement of joints and improve patient quality of life. It is also important to improve gut health and support the nervous system if ongoing stress and emotional trauma are contributing as well as providing topical treatment to help alleviate psoriasis symptoms.
Dietary and lifestyle interventions
Given psoriasis and PsA are highly inflammatory, and have been linked with oxidative stress, implementation of an anti-inflammatory diet that reduces alcohol, sugar and refined foods and is rich in plant-based foods would have significant benefit for patients. The increased fibre as part of an anti-inflammatory diet has also been shown to reduce the inflammatory markers ESR and CRP[14] and will improve bowel function. Additionally, a gluten-free diet is essential given the strong link between PsA and coeliac disease.[15]
Weight management
Obesity is a risk factor for both psoriasis and PsA with several studies showing an increased incidence and severity of these conditions in patients with a body mass index (BMI) >30. Working with patients on weight loss and encouraging them to increase their exercise has been shown to considerably improve symptom severity and treatment outcomes.[12]
Reduce stress
Stress plays an important role as a trigger in both psoriasis and PsA, so managing the stress response is essential.[2] A patient’s lifestyle can markedly affect their stress response, as can managing a chronic condition. So being mindful of time management, good quality and quantity of sleep, sufficient exercise and using coping strategies, such as deep breathing and exercise, will be beneficial.
Safe sun exposure
Ultraviolet (UV) radiation through sun exposure has demonstrated effects in improving psoriasis by rapidly reducing local and systemic inflammatory markers, as well as acting as an immunosuppressant.[16] Over-exposure, causing sunburn, can induce psoriasis flares so regular, incidental sun exposure should be suggested.
Supportive supplements
Curcumin
At 1000mg, curcumin has shown appreciable effects in conditions such as rheumatoid arthritis where inflammation of the joints is a predominant feature. Studies have also found it to be effective in lowering inflammatory markers along with having a lipid lowering effect in patients at risk from developing CVD. These results could suggest curcumin may also be a beneficial treatment for PsA, given the joint involvement, although targeted studies are required to confirm this.[17]
Vitamin D
As PsA is an autoimmune disorder, vitamin D supplementation is an important consideration particularly for those patients on corticosteroids (given this medication reduces vitamin D stores). In addition, vitamin D deficiency is frequently noted in psoriasis and PsA patients. Limited evidence for vitamin D supplementation exists, although doses of 4000IU have been used to address deficiency.[18]
One small trial found great effect with therapeutic doses of 25 hydroxy vitamin D (35,000IU) for the relief of PsA symptoms, but results should be interpreted with caution due to the small trial size.[19]
Fish oil
There is substantial evidence to support the use of omega-3 fatty acids in inflammatory conditions with a minimum of 2g per day being necessary to achieve an anti-inflammatory response.[20] Omega-3 fatty acid supplementation has also shown effect in reducing triglycerides and CVD risk at a minimum dose of 4g daily.[21]
Zinc
Zinc has shown some promise in improving PsA symptoms however studies to-date have been small and have produced conflicting evidence. One study found that 220mg of zinc sulphate given three times daily significantly reduced joint pain and swelling in PsA patients.[22]
In addition, further supplementation may be required, such as folid acid and calcium, depending on medication being used.
Gut testing and management of dysbiosis
Gut microbiome testing provides insight into the presence of dysbiosis, which can be addressed with dietary changes along with pre/probiotic supplementation that should be targeted specifically to each client as needed.
Herbal medicines
Several classes of herbal medicines have shown beneficial effects in the treatment of psoriasis and PsA, although strong evidence in human trials is lacking and more are needed to support documented claims.
Herbal actions that should be considered for these conditions include anti-inflammatories, depuratives, immune modulators, hepatics, digestive aids, antioxidants, adaptogens, adrenal restoratives, analgesics and vulneraries.
Topical treatments for psoriasis in PsA
Topical treatments have had limited success for the treatment of skin lesions in PsA, however some have shown some promise.
A small study found the use of a topical vitamin B12 cream containing avocado oil was effective in reducing psoriasis plaque.[23] Another found that a topical preparation of 0.25% zinc pyrithione cream, applied twice daily, was effective for reducing localised plaque psoriasis.[24] Calcipotriol, a topical vitamin D derivative cream has also shown great effect in reducing pro-inflammatory cytokines that are found in increased levels in psoriatic plaque.[18] Topical vitamin D is thought to be a superior treatment when combined with topical vitamin A.[25]
Additionally, warm bathing in mineral-rich Dead Sea bath salts can aid both skin lesions and joint aches and pains.[26]
Conclusion
PsA is considered a lifelong disease with relapsing and remitting symptoms that can have a significant impact on patient quality of life by affecting their mental, emotional and physical wellbeing. The combination of medications used in the management of PsA whilst effective, can also produce serious side-effects and adverse events leading patients to seek out other supportive treatments.
Naturopathic interventions in the management of PsA including diet, lifestyle, herbs and supplements have proven to be safe and effective. However, it is a complicated disease that can include several comorbid conditions and as a result, is likely to require an extensive treatment regime.
Several of the studies supporting the use of herbs, supplements and lifestyle interventions in PsA, whilst demonstrating positive outcomes, are small and further research is needed to confirm their effectiveness on a wider population. Nevertheless, the use of targeted interventions and a more holistic approach to the treatment of PsA that addresses all contributing factors in this condition can help to reduce symptoms and improve patient quality of life.
References
- Celgene Corporation. Global Burden of Psoriasis Recognized by WHO 2014. [Source]
- Chandran V, Raychaudhuri SP. Geoepidemiology and environmental factors of psoriasis and psoriatic arthritis. J Autoimmunity 2010;34(3):J314-J321. [Abstract]
- Wilsdon TD, Whittle SL, Thynne, et al. Methotrexate for psoriatic arthritis (Protocol). Cochrane Database of Systematic Reviews 2017;2017(7):1-13. [Full Text]
- Deng S, Cheng J, Zhao J, et al. Natural compounds for the treatment of psoriatic arthritis: A proposal based on multi-targeted osteoclastic regulation and on a preclinical study. JMIR Res Protocols 2017;6(7):e132. [Full Text]
- Liu JT, Yeh HM, Liu SY, et al. Psoriatic arthritis: Epidemiology, diagnosis, and treatment. World J Orthopedics 2014;5(4):537-543. [Full Text]
- Busse K, Liao W. Which psoriasis patients develop psoriatic arthritis?. Psoriasis forum / National Psoriasis Foundation 2010;16(4):17-25. [Full Text]
- Moll J, Wright V. Psoriatic arthritis. Seminars in Arthritis and Rheumatism 1973;3(1):55-78. [Abstract]
- Simonić E, Peternel S, Stojnić-Soša L, et al. Negative and positive life experiences in patients with psoriatic arthritis. Rheumatol International 2013;33(6):1587-1593. [Abstract]
- Sankowski AJ, Lebkowska UM, Jaroslaw C, et al. Psoriatic arthritis. Polish J Radiology 2013;78(1):7-17. [Full Text]
- Popescu C, Pintilie AM, Bojinca V, et al. Cardiovascular risk in psoriatic arthritis - a cross-sectional study. Maedica 2014;9(1):19-24. [Full Text]
- Lindqvist U, Rudsander Å, Boström Å, et al. IgA antibodies to gliadin and coeliac disease in psoriatic arthritis. Rheumatology 2002;41(1):31-37. [Full Text]
- Clemente JC, Manasson J, Scher JU. The role of the gut microbiome in systemic inflammatory disease. BMJ 2018;8(5):1-16. [Abstract]
- Mahmood F, Helliwell P. Psoriatic arthritis: A review. European Med J 2016;3(1):114-117. [Full Text]
- Ajani U, Ford E, Mokdad A. Dietary fiber and C-reactive protein: findings from National Health and Nutrition Examination Survey Data. J Nutr 2004;134(5):1181-1185. [Full Text]
- Bhatia BK, Millsop JW, Debbaneh M, et al. Diet and psoriasis: Part 2. celiac disease and role of a gluten-free diet. J American Academy Dermatol 2014;71(2):350-358. [Full Text]
- Søyland E, Heier I, Rodríguez-Gallego C, et al. Sun exposure induces rapid immunological changes in skin and peripheral blood in patients with psoriasis. BJD 2011;164(2):344-355. [Abstract]
- Amalraj A, Varma K, Jacob J, et al. A novel highly bioavailable curcumin formulation improves symptoms and diagnostic indicators in rheumatoid arthritis patients: A randomized, double-blind, placebo-controlled, two-dose, three-arm, and parallel-group study. J Medicinal Food 2107;20(10):p. jmf.2017.3930. [Abstract]
- Barrea L, Savanelli MC, Di Somma C, et al. Vitamin D and its role in psoriasis: An overview of the dermatologist and nutritionist. Rev Endocrine Metabolic Dis 2017;18(2):195-205. [Full Text]
- Finamor DC, Sinigaglia-Coimbra R, Neves LCM, et al. A pilot study assessing the effect of prolonged administration of high daily doses of vitamin D on the clinical course of vitiligo and psoriasis. Dermato-Endocrinology 2013;5(1):222-234. [Full Text]
- Calder PC. Omega-3 polyunsaturated fatty acids and inflammatory processes: Nutrition or pharmacology? British J Clinical Pharmacol 2013;75(3):645-662. [Full Text]
- Weitz D, Weintraub H, Fisher E, et al. Fish oil for the treatment of cardiovascular disease. Cardiology Rev 2010;18(5):258-263. [Full Text]
- Clemmensen OJ, Siggaard-Andersen J, Worm AM. Psoriatic arthritis treated with oral zinc sulphate. BJD 1980;103(4):411-415. [Abstract]
- Stücker M, Hoffmann M. Vitamin B 12 cream containing avocado. Dermatology 2001: 141-147. [Abstract]
- Gupta M, Mahajan VK, Mehta KS, et al. Zinc therapy in dermatology: A review. Dermatol Res Practice 2014;2014:1-11. [Full Text]
- Usedom E, Neidig L, Allen H. Psoriasis and fat-soluble vitamins: A review. J Clinical Experimental Dermatol Res 2017;8():1-6. [Full Text]
- Halevy S, Giryes H, Friger M, et al. The role of trace elements in psoriatic patients undergoing balneotherapy with Dead Sea bath salt. Israel Medical Association Journal 2001;3(11):828-832. [Full Text]
DISCLAIMER:
The information provided on FX Medicine is for educational and informational purposes only. The information provided on this site is not, nor is it intended to be, a substitute for professional advice or care. Please seek the advice of a qualified health care professional in the event something you have read here raises questions or concerns regarding your health.