Anxiety disorders appear to be caused by an interaction of biopsychosocial factors, including genetic vulnerability, which interact with situations, stress or trauma to produce clinically significant syndromes. Symptoms vary depending on the specific anxiety disorder.[1]
Anxiety becomes a disorder when it is irrational, excessive, too persistent or attached to situations that do not warrant it and interferes with the person’s daily life.
Anxiety conditions include:
- generalised anxiety disorder (GAD)
- social phobias, fear of social situations
- specific phobias, for example fear of open spaces (agoraphobia) or enclosed spaces (claustrophobia)
- panic disorder, frequent and debilitating panic attacks
- obsessive compulsive disorder (OCD)
- post-traumatic stress disorder (PTSD).
Generalised anxiety is excessive anxiety and worry about common issues. The affected person finds the worries difficult to control, and this can result in decreased occupational and social functioning.[2,3]
The focus of the anxiety might be family, friends, health, work or money. Even minor things, such as household chores or being late for an appointment, can become the focus of the anxiety, leading to uncontrollable worries and a feeling that something terrible will happen.[2,3]
GAD commonly coexists with other anxiety and depressive disorders, as well as a variety of physical health conditions. Symptoms include: irritability, poor concentration, increased sensitivity to noise, sleep disturbance (typically difficulty falling asleep), sweating, dry mouth, palpitations, urinary frequency, epigastric discomfort and frequent and/or loose bowel motions, increased muscle tension, inability to relax, headaches and aching pains, particularly in the shoulders and back.[2]
A person may be diagnosed with a GAD if:[3,4]
- the anxiety and worry has been present most days over the last six-months
- the person finds it difficult to control their anxiety.
Every year in Australia, about 14% of the population (1 in 7) experience an anxiety condition and it is estimated that just under 3% experience GAD. Nearly 6% of the population will experience GAD in their lifetime.[4]
The condition tends to affect more women than men. It can occur at any time in life and is common in all age groups, including children and older people, although on average it starts around 21-30 years of age. The average duration of the condition is 20 years.[2,4]
The majority of people with GAD do not seek medical help for their symptoms. Although, since GAD also often co-occurs with physical health problems, such as arthritis and gastrointestinal and respiratory disorders, and may mimic the presentation of some physical conditions (e.g. hyperthyroidism), these people usually seek (and receive) help for the physical symptoms rather than the emotional symptoms, with the diagnosis of GAD being missed.[2]
What causes GAD?
People with GAD often have a history of mental health problems in their family. Overall findings suggest that genetic factors play a significant, though moderate, role in the aetiology of GAD. Genetic factors predispose people to a range of anxiety and depressive disorders rather than GAD specifically. Whereas, environmental factors are important in determining the nature of the emotional disorder experienced by a particular person.[2]
The brain circuits and regions associated with anxiety disorders are beginning to be understood, with the development of functional and structural imaging. The brain amygdala appears key in modulating fear and anxiety. Patients with anxiety disorders often show heightened amygdala response to anxiety cues. Hyper-responsiveness of the amygdala may relate to reduced activation thresholds when responding to perceived social threat.[1]
People may be more at risk if they experience a major life change that causes stress, such as the birth of a child, the breakdown/loss of a close relationship or moving house/job. Physical, sexual or emotional abuse also increase the risk of developing GAD, as do other traumatic experiences in childhood such as the death of, or separation from, a parent. Some individuals appear resilient to stress and others more vulnerable to stress, which can precipitate an anxiety disorder.[1,4]
Some personality traits may put a person at greater risk of GAD including being sensitive, emotional or experiencing general nervousness, inability to tolerate frustration, feeling inhibited, lacking a sense of control and having perfectionistic tendencies.[4]
Such individuals may also demonstrate increased attention to (perceived) potentially threatening stimuli, overestimation of an environmental threat and enhanced memory of threatening material. Studies suggest that people at risk of GAD use worry as a positive coping strategy to deal with potential threats, whereby the person worries until they feel reassured that they have appraised all possible dangers and identified ways of dealing with them. These individuals come to believe that worrying in this way is necessary for them, but it is actually uncontrollable and harmful.[2]
Biochemistry and underlying pathology
The biochemistry of anxiety is complex. Nearly every type of neurotransmitter and hormone can play some role in anxiety, as can anything that reduces blood flow to the brain (like dehydration). Having too little of the inhibitory neurotransmitters (GABA, dopamine and serotonin) or too much of the excitatory neurotransmitters (adrenaline, noradrenaline and glutamate) has been linked to anxiety disorders. Suppressed brain-derived neurotrophic factor (BDNF) is also observed in patients with depression and certain anxiety disorders, particularly obsessive compulsive disorder (OCD).
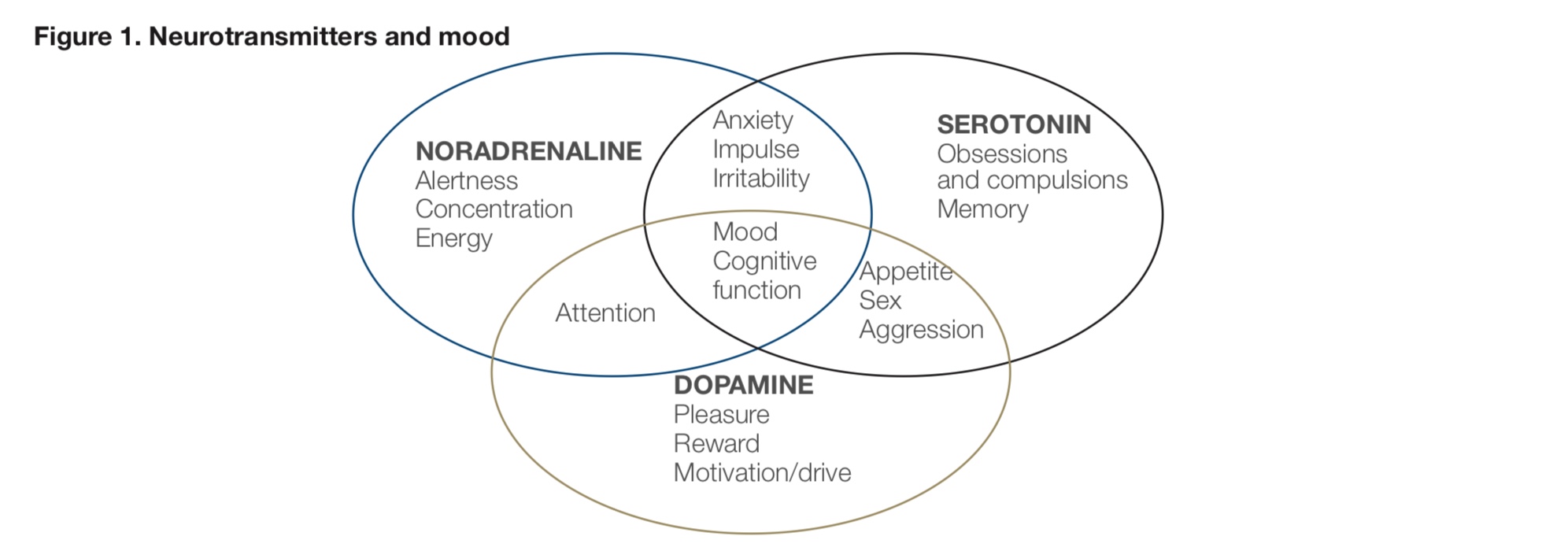
In the central nervous system (CNS), serotonin is believed to play an important role in the regulation of anger, appetite, body temperature, mood, sexuality and sleep. Low levels may be associated with aggression, anxiety, depression, eating disorders, impulsivity, irritability and sleep disorders.
Serotonin is formed from the essential amino acid tryptophan. In the first step to produce serotonin, tryptophan is hydroxylated to form 5-hydroxytryptophan (5-HTP), which is then converted to serotonin. Serotonin can then be metabolised by two different biochemical pathways. It can be converted to melatonin, a reaction which predominantly occurs in the pineal gland. This pathway requires an enzyme known as COMT which uses S-adenosylmethionine (SAMe) and magnesium as cofactors. The other pathway breaks down serotonin in the body and utilises the enzyme MAO-A; a reaction in which 5-hydroxyindoleacetic acid may be reduced and a side pathway making 5-hydroxytryptopphol (5HTOL) may be favoured.
Serotonin and dopamine interact with each other; if one goes up, the other often goes down.
Dopamine is an excitatory and inhibitory neurotransmitter synthesised in many areas of the brain. It is the precursor for noradrenaline and adrenaline. Dopamine also acts as a hormone when it is released from the hypothalamus, inhibiting prolactin production from the pituitary gland. In the CNS, dopamine is involved in the regulation of pleasure and reward, memory, motor control, sleep, mood, attention and learning. Dopamine is released by rewarding experiences such as food, sex and (some) drugs. Lowered dopamine has been associated with loss of satisfaction, social withdrawal, apathy, reduced motivation and attention. In addition, low dopamine levels can result in impaired motor control, e.g. Parkinson’s disease. High levels of dopamine may result in aggression, schizophrenia, hyperactivity and Tourette’s syndrome.
Noradrenaline and adrenaline are excitatory neurotransmitters, as well as hormones. They are produced by noradrenergic and adrenergic neurons respectively, as well as by the adrenal medulla. They are most known for their involvement in the ‘fight and flight’ response, in which they increase heart rate, trigger the release of glucose from energy stores and increase blood flow to skeletal muscle. Low levels contribute to decreases in mood, energy, focus, motivation and memory. High levels are associated with aggression, anxiety, emotional lability, hyperactivity, mania, stress and suppression of the immune system.
Gamma aminobutyric acid (GABA) is the brain’s major inhibitory neurotransmitter. When GABA binds to a GABA receptor in the brain, it causes a reduction in the ability of that neuron to conduct a neural impulse. Thus, GABA has the ability to “shut down” nerve cells throughout the CNS.
Glutamate is excitatory and binds at the N-methyl-D-aspartate (NMDA) receptor. It is the precursor to GABA and is important in learning and memory. Excess glutamate may contribute to excitotoxicity, suppressing BDNF. Interestingly, zinc and magnesium are identified as interacting with, and acting as an antagonist at the NMDA receptor, potentially minimising the negative consequences of elevated glutamate.
Brain-derived neurotrophic factor (BDNF) is a secretory growth factor that supports the survival of existing neurons and promotes synaptogenesis and differentiation of new neurons. It is a neurotrophin essential for neuronal development and survival, synaptic plasticity and cognitive function. Dysregulation of BDNF signalling is involved in several mood and neurodegenerative disorders, including depression and Alzheimer's.
Contributing gene variations
Genetic factors can influence the risk for many anxiety disorders and the following genes should be reviewed.
Monoamine oxidase A (MAO-A)
MAO-A is an enzyme that metabolises the neurotransmitters dopamine, serotonin, adrenaline and noradrenaline as well as xenobiotic amines. Variations can result in both high and low activity. High activity may see low monoamines and low mood, whilst low activity can contribute to neurotransmitter accumulation that may present as anxiety, aggression or social withdrawal.
Phosphatidylethanolamine N-methyltransferase (PEMT)
PEMT converts phosphatidylethanolamine (PE) to phosphatidylcholine (PC) and is required for the synthesis of choline. The SNP may reduce PC and choline, compromising cell membrane, neurological and liver health. Patients presenting with this variation may benefit more from krill oil and/or PC supplementation than fish oil.
Brain-derived neurotrophic factor (BDNF)
BDNF is involved in cognitive function and protecting and regenerating neurons. A SNP on the gene coding for BDNF can reduce levels, increasing risk of mood disorders, dementia and migraines. Exercise, zinc, magnesium, curcumin and resveratrol may promote BDNF expression.
Tyrosine hydroxylase (TH)
TH converts L-tyrosine into L-DOPA, which in-turn can be converted into noradrenaline and adrenaline, and then onto dopamine. The L-DOPA to dopamine conversion requires aromatic amino acid decarboxylase, which is dependent on vitamin B6. The SNP can increase TH activity, increasing levels of neurotransmitters and risk of anxiety and high blood pressure.
Catechol-o-methyl transferase (COMT)
COMT metabolises/inactivates catecholamines, including the neurotransmitters dopamine, adrenaline and noradrenaline as well as catechol oestrogens. Certain SNPs can reduce activity, potentially leading to higher levels of neurotransmitters, which may express itself as anxiety. Being a methyltransferase, COMT requires SAMe. Therefore B vitamins may help, as well as magnesium.
Glutamic acid decarboxylase 1 (GAD1)
GAD1 converts glutamate to GABA. A SNP on this gene results in lower activity which increases the risk of mood disorders and poorer working memory. As GABA is a known inhibitory neurotransmitter, which helps to “switch off” the stress response, a reduction in the availability of this neurotransmitter can contribute to anxiety. Additionally if glutamate levels remain elevated, this can also contribute to anxiety and excitotoxicity. High levels of stress hormones can see glutamate accumulate, and high levels of glutamate are excitotoxic and are linked to neurodegenerative diseases.
Zinc and magnesium each act as an antagonist of glutamate at the NMDA receptor, while vitamin B6 is essential to support healthy glutamic acid decarboxylase function. Certain herbs (e.g. magnolia and phellodendron) contain actives that act as an agonist at the GABA receptor, helping it to function and may also be indicated here. In patients with elevated pyrroles, the effects of this genetic variation may be compounded by the resultant low levels of vitamin B6 and zinc.
The brain has three types of GABA receptors: GABAA, GABAB and GABAC. GABAA receptors mediate fast inhibitory synaptic transmissions. They regulate neuronal excitability, such as the seizure threshold, and rapid mood changes, such as panic. GABAA receptors are the targets of sedating drugs, such as benzodiazepines, barbiturates, neurosteroids and ethanol. GABAB receptors mediate slow inhibitory potentials. They play an important role in memory, depressed moods and pain. Stimulation of GABAB receptors can also reduce the release of dopamine, thereby inhibiting the reward/reinforcing response to drug abuse. GABAC receptors are found in the retina; their physiologic role is poorly understood.
D-amino acid oxidase (DAO)
Also known as diamine oxidase, DAO is involved in the degradation of histamine and D-amino acids such as D-serine, D-alanine, D-proline and D-leucine. SNPs resulting in low functioning may mean the patient is more prone to migraines and anxiety because DAO is involved in glutamate receptor function. DAO is dependent on vitamins B6, B12, iron, copper and vitamin C.
GC-globulin (GC)
GC codes for the vitamin D binding protein (DBP). DBP is the primary vitamin D transporter and plays a role in maintaining the total levels of vitamin D and regulating the amounts of free (unbound) vitamin D available for specific cells and tissues. The SNP reduces function and may increase risk of vitamin D deficiency and vitamin D deficiency is linked to depression.
CYP2R1
This SNP codes for an enzyme that converts vitamin D obtained from sunlight, the diet and supplements into 25-hydroxyvitamin D (25-OH D). 25-OH D is the major circulating form of vitamin D and acts as a precursor for the active form of vitamin D. The SNP results in greater risk of vitamin D deficiency and vitamin D deficiency is linked to depression.
Methylenetetrahydrofolate reductase (MTFHR)
MTHFR converts 5,10-methylenetetrahydrofolate (5,10-MTHF) to 5-methyltetrahydrofolate (5-MTHF). 5-MTHF is the form of folate that can give a methyl group to homocysteine to make methionine that is then used to synthesise SAMe. The SNP can reduce its function, resulting in potentially higher homocysteine and lower methionine and SAMe. Since COMT function and conversion of serotonin to melatonin require SAMe, depleted SAMe can mean an increase in the levels of stimulating neurotransmitters and therefore anxiety.
Serine hydroxymethyltransferase 1 (SHMT1)
SHMT1 converts serine and THF to glycine and 5,10 methylene-THF. Low function due to the SNP will reduce active folate, resulting in increased homocysteine and possible problems with DNA synthesis.
Pathology considerations
- Zinc (or copper:zinc ratio) and magnesium
- Mauve factor/HPL/pyrroles
- Extensive neurotransmitter profile
- Histamine - indication of over/under methylation
- Vitamin D (25-hydroxyvitamin D)
- Vitamin B2, B6, folate, B12 and homocysteine
- Iron studies - iron deficiency is linked to an increased risk of depression
Treatment considerations
There are two main types of conventional treatments used for GAD: psychological treatments will generally be the first line of treatment; and, in more severe cases, medication can also be effective.
Referral to a psychologist is strongly suggested with GAD, or a psychiatrist for more severe cases where medication may be required.
Other modalities of assistance may include:
- exercise
- yoga
- prayer/religion
- relaxation techniques
- cognitive behavioural therapy
- mindfulness practise.
Nutritional and herbal supplementation are also important for the anxious patient and can greatly support and assist their ability to cope.
- Magnesium – deficiency is common amongst those with anxiety and other mood disorders.
- Zinc – deficiency is noted in depression, and concurrent zinc and antidepressant medication improves drug effectiveness.
- Active folate – supports healthy methylation, an essential step in neurotransmitter synthesis and metabolism. Folate deficiency is identified in approximately one third of depressed individuals and many more are thought to exhibit an insufficiency, making it’s supplementation useful. Improving folate status may also improve the success of antidepressant medication.
- Amino acids (serine, methionine, phenylalanine, tyrosine) and B vitamins (folate, vitamins B6, B2 and B12) are essential in the synthesis of multiple neurotransmitters (serotonin, dopamine, melatonin, adrenaline) and essential phospholipids (phosphatidylcholine).
- Vitamin B6 – involved in conversion of 5-hydroxytryptophan (5-HTP) to 5-HT (serotonin) and also glutamate to GABA.
- SAMe – supports methylation, assists synthesis of “mood lifting” neurotransmitters, and addresses common deficiencies linked with depression and anxiety.
- EPA and DHA – essential for cell membrane health, neurotransmission and healthy glucose metabolism in neuronal cells. A deficiency of these EFAs is linked to an increased risk of depression and defective neurotransmission. It is believed EPA may exert its therapeutic effects also through reduction of cortisol. DHA is essential in the health and structure of the brain and neural networks.
- Vitamin C – essential for the functioning of white blood cells for immune function and is commonly depleted in chronically stressed individuals.
- Herbal supplementation would also be beneficial for various symptoms of anxiety – kava, withania, Siberian ginseng, lemon balm, holy basil, motherwort, chamomile, valerian, magnolia, medicinal mushrooms.
References
- Bhatt NV. Anxiety disorders. Medscape 2017. [Source]
- National Collaborating Centre for Mental Health (UK). Generalised Anxiety Disorder in Adults: Management in Primary, Secondary and Community Care. Leicester (UK): British Psychological Society; 2011. (NICE Clinical Guidelines, No. 113.). [Full Text]
- Better Health Channel. Anxiety disorders. Victoria State Government 2017. [Source]
- Beyond Blue. GAD. Beyond Blue 2018. [Source]
DISCLAIMER:
The information provided on FX Medicine is for educational and informational purposes only. The information provided on this site is not, nor is it intended to be, a substitute for professional advice or care. Please seek the advice of a qualified health care professional in the event something you have read here raises questions or concerns regarding your health.



