What is methylation?
No topic has grabbed the attention of integrative health practitioners quite like methylation has. One of the reasons for this is that imbalances in methylation pathways are extremely common and may underpin a plethora of health conditions ranging from cardiovascular disease to depression. Discovering and treating these imbalances may provide the key to treating certain complex cases.
So what exactly is methylation? Methylation is a fundamentally important biochemical process whereby a methyl group, consisting of one carbon and three hydrogen atoms, is transferred to a substrate such as DNA, RNA, neurotransmitters, hormones, immune cells or nerve cells. These newly methylated compounds can then complete their tasks in the body, which includes detoxification, DNA and RNA synthesis and repair, gene regulation, neurotransmitter production, hormone regulation, energy production, cell membrane repair, fat metabolism, myelination, and immune function.[1] When methylation is impaired these processes become compromised and ill health may arise. Methylation occurs billions of times every second and involves several pathways including the folate and methionine cycles.
Methylation imbalance - what can go wrong?
The folate and methionine cycles involved in methylation are highly dependent on an adequate supply of several key nutrients which act as cofactors and substrates. Without an adequate supply of these nutrients, which include folate, vitamin B2, vitamin B6, vitamin B12, serine and choline, methylation imbalances can occur. Inadequate dietary intake of these nutrients, nutrient malabsorption and medications that deplete nutrient levels can cause methylation imbalances. Single nucleotide polymorphisms (SNPs) including MTHFR polymorphisms are also a significant contributor to methylation imbalances (see breakout box). Lifestyle factors such as chronic alcohol intake, cigarette smoking, stress, and environmental toxicity and exposure can also result in methylation imbalances.[2,3]
Methylation imbalances result in a build-up of homocysteine and inadequate production of methionine and S-adenosylmethionine (SAMe) which is the major methyl donor in the body. The consequences of high homocysteine, inadequate SAMe and defective methylation include vascular damage, increased oxidative stress, inflammation, damage to DNA and dysregulation of DNA repair, neurotoxicity, altered neurotransmitter metabolism, reduced detoxification and reduced endogenous antioxidant production. This may translate into ADD/ADHD, allergies, autism spectrum disorders, bone fractures, cardiovascular disease, cancer, chronic viral infections, cognitive decline, diabetes, immune dysfunction, fertility issues, macular degeneration, migraine, mood disorders (including anxiety and depression), multiple sclerosis, neural tube defects, neuropathy, pregnancy complications and thyroid dysfunction.[3-6]
The importance of healthy methylation support – folate
The key nutrient for supporting methylation, and the folate and methionine cycles, is folate, also known as vitamin B9. Before incorporation into the folate cycle, folic acid undergoes enzymatic modification at several key steps to be transformed into dihydrofolate (DHF), then tetrahydrofolate (THF), then 5,10-methylene-THF and ultimately 5-methyl-THF (5-MTHF), the active form of folate. 5-MTHF is then able to donate its methyl group to homocysteine transforming it into methionine (see Figure 1).
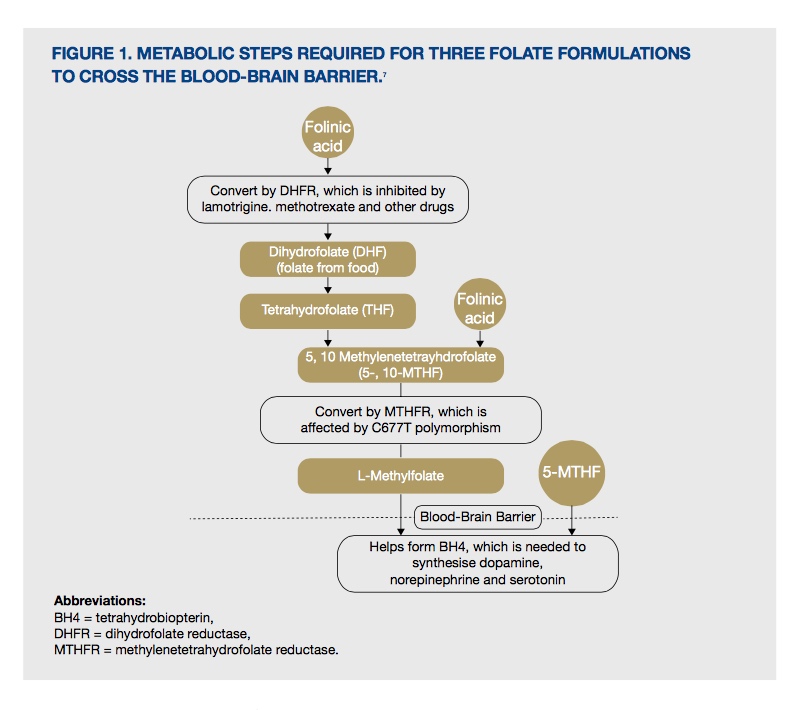
Figure 1. Metabolic steps required for three folate formulations to cross the blood-brain barrier.[7]
The transformation of folate, or folic acid, into 5-MTHF is dependent on the activity of the enzymes dihydrofolate reductase (DHFR) and methylenetetrahydrofolate reductase (MTHFR). An emerging health problem is the discovery of the under functioning of these enzymes due to genetic SNPs or medication use, which affect an individual’s ability to metabolise folic acid. Up to 45-50% of the general population may be affected by a MTHFR gene variance, which may reduce their ability to produce active folate thus directly impairing methylation in the body.[1]
Forms of supplemental folate
Supplemental folate is available in three main forms, folic acid, folinic acid and 5-MTHF, and their use is determined by an individual’s health status and functional enzyme capacity.
Folic acid
Folic acid was the first and original supplemental form of folate used, due to its stability and high absorption rate. However, folic acid is not a coenzyme form of folate and must be reduced to become metabolically active, which is dependent on the DHFR and MTHFR enzymes and an adequate supply of cofactors, including vitamin B2, vitamin b6, zinc and serine.[8] Concerns have been raised surrounding the presence of unmetabolised folic acid in the bloodstream following folic acid supplementation and food fortification, and the possible negative effects this has on the body.[9]
Folinic acid
Folinic acid is a more metabolically active form of folate, which does not require the enzyme DHFR for activation. Folinic acid has several advantages over folic acid as it is more easily transported into the CNS, has a longer half-life, and is able to increase coenzyme forms of folate where folic acid has had minimal or no effect.[8] As it does not require DHFR for activation, folinic acid is not affected by medications which inhibit DHFR function, including methotrexate, pyrimethamine and trimethoprim.[10]
5-MTHF
One of the most exciting nutrients recently made available for Australian practitioners is 5-MTHF. 5-MTHF is the active form of folate and the main form of circulating folate. In regard to methylation, 5-MTHF donates a methyl group to vitamin B12, which subsequently transfers it to homocysteine, which is then converted to methionine (see Figure 2).[11] Without an adequate supply of 5-MTHF, homocysteine levels may become elevated and methionine production decreased.
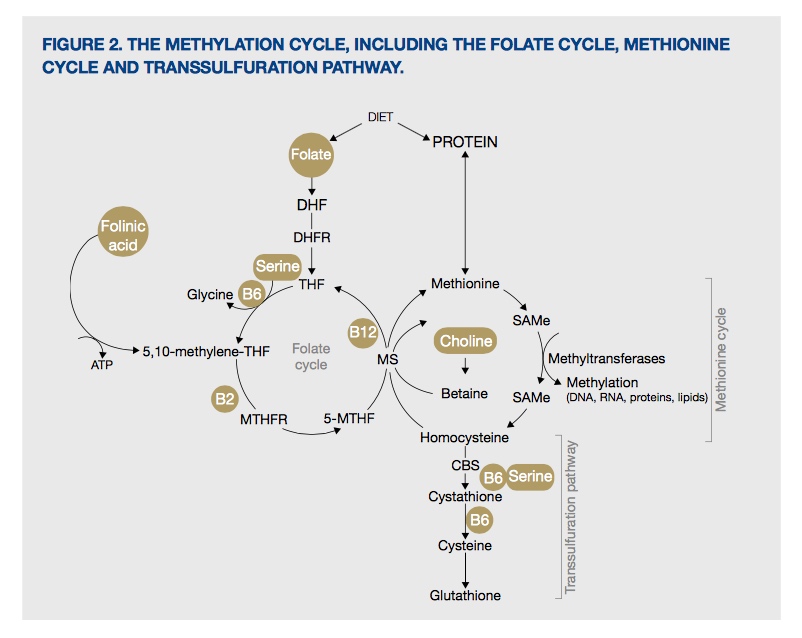
Figure 2. The methylation cycle, including the folate cycle, methionine cycle and transsulfuration pathway
As 5-MTHF provides folate in the ‘last step’ of the folate cycle, it does not require the enzymes DHFR or MTHFR. This is of particular importance, and of potential benefit, to individuals with MTHFR polymorphisms (and potentially DHFR polymorphisms although this research is still in its infancy), as 5-MTHF provides a direct source of active folate for methylation (see breakout box).
So which form of supplemental folate to prescribe?
As 5-MTHF and folinic acid are ‘newer’ supplemental forms of folate, their use in clinical trials is not as extensive as the ‘older’ form folic acid. However, research shows that supplementation with 5-MTHF increases blood folate levels equally to folic acid supplementation, and may even be more effective.[12-14] Research confirms that 5-MTHF and folinic acid supplementation increases serum folate levels, improves folate status and reduces homocysteine levels, and appears to provide advantages over folic acid.[10]
Researchers are increasingly recommending the use of 5-MTHF or folinic acid as alternatives to folic acid for both supplementation and food fortification.[10] Individuals with methylation defects are most likely to benefit from 5-MTHF or folinic acid supplementation over folic acid, where these active forms of folate can be incorporated directly into the methylation pathways, without the need for activation.[11]
Supporting nutrients for healthy methylation
Like any biochemical pathway in the body, methylation requires a synergistic group of nutrients to support each step in the interconnected folate and methionine cycles. In addition to folate, the body requires vitamin B2, B6, B12, choline and serine, which act as cofactors or methyl donors.
References
- MTHFR Support Australia. Frequently asked questions. [Link]
- Lynch B. Methylation and MTHFR defects, 2012. [Link]
- Miller AL. The methionine-homocysteine cycle and its effects on cognitive diseases. Altern Med Rev 2003;8(1):7-19. [Full Text]
- MTHFR Support Australia. Conditions. [Link]
- Kennedy DO. B Vitamins and the brain: mechanisms, dose and efficacy — a review. Nutrients 2016;8(2):68. [Full Text]
- Gopinath B, Flood VM, Rochtchina E, et al. Homocysteine, folate, vitamin B-12, and 10-y incidence of age-related macular degeneration. Am J Clin Nutr 2013;98(1):129-135. [Full Text]
- Fava M, Mischoulon D. Folate in depression: efficacy, safety, differences in formulations, and clinical issues. J Clin Psychiatry 2009;70 Suppl 5:12-17. [Full Text]
- Kelly GS. Folates: supplemental forms and therapeutic applications. Altern Med Rev 1998;3(3):208-220. [Full Text]
- Prinz-Langenohl R, Brämswig S, Tobolski O, et al. [6S]-5-methyltetrahydrofolate increases plasma folate more effectively than folic acid in women with the homozygous or wild-type 677C-->T polymorphism of methylenetetrahydrofolate reductase. Br J Pharmacol 2009;158(8):2014-2021. [Full Text]
- Scaglione F, Panzavolta G. Folate, folic acid and 5-methyltetrahydrofolate are not the same thing. Xenobiotica 2014;44(5):480-488. [Full Text]
- 5-methyltetrahydrofolate monograph. Altern Med Rev 2006;11(4):330-337. [Full Text]
- Venn BJ, Green TJ, Moser R, et al. Comparison of the effect of low-dose supplementation with L-5-methyltetrahydrofolate or folic acid on plasma homocysteine: a randomized placebo-controlled study. Am J Clin Nutr 2003;77(3):658-662. [Full Text]
- Lamers Y, Prinz-Langenohl R, Brämswig S, et al. Red blood cell folate concentrations increase more after supplementation with [6S]-5-methyltetrahydrofolate than with folic acid in women of childbearing age. Am J Clin Nutr 2006;84(1):156-161. [Full Text]
- Houghton LA, Sherwood KL, Pawlosky R, et al. [6S]-5-Methyltetrahydrofolate is at least as effective as folic acid in preventing a decline in blood folate concentrations during lactation. Am J Clin Nutr 2006;83(4):842-850. [Full Text]
- Gilbody S, Lewis S, Lightfoot T. Methylenetetrahydrofolate reductase (MTHFR) genetic polymorphisms and psychiatric disorders: a HuGE review. Am J Epidemiol 2007;165(1):1-13. [Full Text]
DISCLAIMER:
The information provided on FX Medicine is for educational and informational purposes only. The information provided on this site is not, nor is it intended to be, a substitute for professional advice or care. Please seek the advice of a qualified health care professional in the event something you have read here raises questions or concerns regarding your health.



